In response to analyses of H
2 of some
25,000ppm reported by Landis and Hofstra in their paper "
Ore
genesis constraints on the Idaho cobalt belt from fluid
inclusion gas, noble gas isotope and ion ratio analyses",
Economic Geology Sept.-Oct. 2012, V107, #6, P1189, I submitted a
discussion paper to query the reliability of analysis of H
2
by mass spectrometry. This was published in Economic Geology
August 2013, V108 #5 p1211, together with a reply from Landis
and Hofstra.
This
discussion and reply is here (as a pdf file).
The intent of the discussion was to bring attention to many
unexplained or incorrectly explained analyses of hydrogen within
aqueous analytes by mass spectrometry (MS) and to suggest that
confirmation analyses using a more robust analytical technique
such as Laser Raman analyses should be performed before relying
on mass spectrometric data for H
2.
Although there may actually be H
2 in the fluids
analysed by Landis and Hofstra, they have chosen not to provide
independent verification by Laser Raman Spectroscopy and the
issue of unexpected and potentially erroneous MS analyses of H
2
reported by many authors has not been dealt with. The problem of
suspiciously high H
2 results by MS was identified by
D. Norman and F. Sawkins (
Analysis
of volatiles in fluid inclusions by mass spectrometry,
Chemical Geology, V61, #4, March 1987) who suggested that the H
2
was the product of reaction of CH
4 and H
2O
within the inclusions during analysis. But this reaction is
thermodynamically improbable
as previously discussed here.
Landis and Hofstra provide confirmation in their reply that this
chemical reaction is not the cause of high H
2
results. Unfortunately this erroneous proposed mechanism seems
to have been accepted and prevented proper further evaluation of
the H
2 analytical problem to find the real cause of
the spurious results.
Landis and Hofstra refer to "background" levels of H
2
in the mass spectrometer. This is precisely the problem. Why
should there be any background H
2 in the
spectrometer? Why do residual gas analyses of supposedly empty
vacuum chambers ( shown
here, and
here,
and
here)
consistently show high levels of H
2 at mass 2 when we
are quite sure that H
2 is not a background gas in the
atmosphere? It seems that it is assumed that H
2 comes
from the walls of the instrument, but surely this is an
obfuscation to avoid the absence of a proper explanation. Note
that despite the very
careful
analysis of the cause of all the peaks in this spectrogram,
there is no explanation of the peak at mass 2 for H
2,
merely a statement that it is caused by hydrogen. But why should
there be any hydrogen in residual atmospheric air? (The website
owners have not responded to a request for an explanation.)
The extremely high analyses of H
2 by mass
spectroscopy in fluid inclusion volatiles in this sample of
proterozoic quartz is
also discussed here.
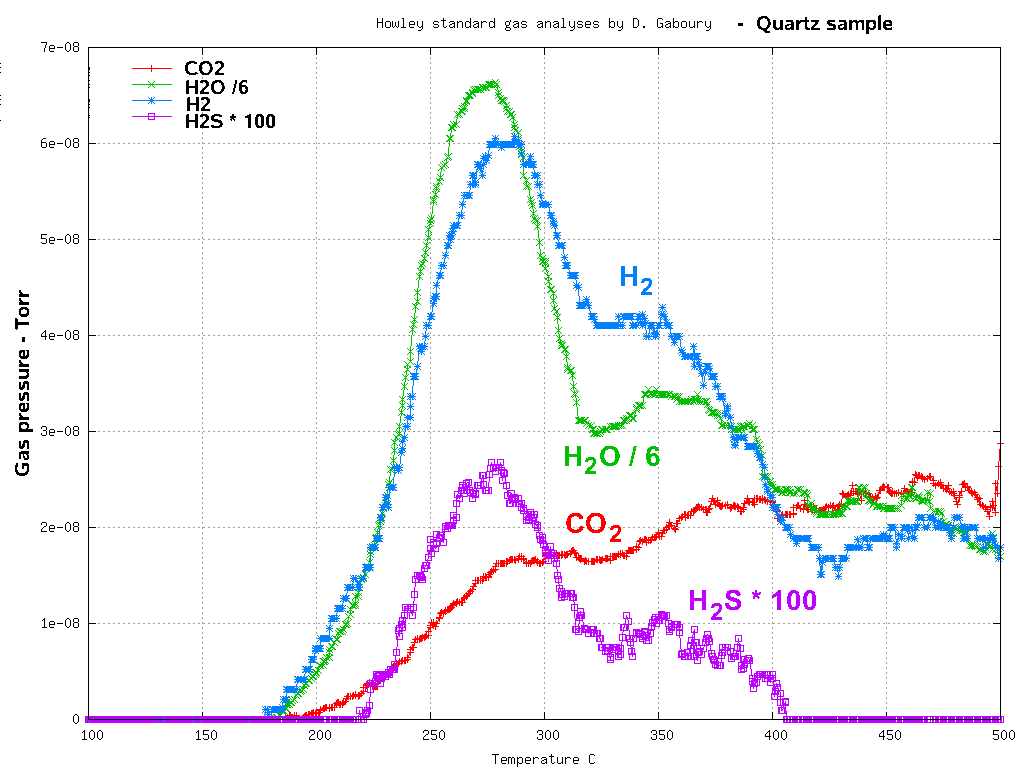
The proterozoic aged quartz sample analysed here was collected
from surface outcrop as hand sized fragments in 1980 and crushed
to <400 micron size and stored with only a thin plastic cover
without even being airtight. The approximately 30 years of
storage under atmospheric conditions alone is enough for
diffusion to seriously deplete any hydrogen which may have been
present in the fluid inclusions. It is inconceivable that the H
2
reported in this analysis, supposedly even more than the CO
2,
originated from the trapped inclusion volatiles.
These problems are indicative of an unexplained instrumental
issue. Landis and Hofstra dismiss this as "fundamental mass
spectrometry", but I suggest that there is indeed a problem with
fundamental mass spectrometry when the presence of water in the
analyte is ignored. Landis and Hofstra state that they "
analyze
mixtures of individual gas species with N2
or Ar to document their relative ionization efficiencies and
fragmentation in the source of our quadrupole mass
spectrometer." But they do not mention if this is done in
a vacuum system saturated with water, which is the normal
condition for fluid inclusion volatiles analysis.
As discussed here, the
presence of water in the vacuum could cause significant
interference with H
2 analyses. Correction
coefficients determined in a dry vacuum may not be appropriate
for analyses under wet conditions because of H
+ ion
impacts with the H
2O adsorbed on all the apparatus
electrodes, potentially generating H
2 from water
alone.
Summary
Mass spectrometric analysis
of H2 in fluid inclusions has long been recognized
as problematical by various authors. Analyses of residual gas
in mass spectrometer vacuums also routinely shows the presence
of H2 at mass 2, but is only explained as
"background" when it is unclear why there should be any
background of H2 in residual atmospheric air. I
suggest that the problem is caused by the generation of H2
from water in the electron impact ionizer as explained here. It is incorrect to
merely ignore the presence of H2O adsorbed onto the
electrode surfaces of the instrument. Positive ions from the
ionizer are attracted to these water coated electrode
surfaces, which are negatively charged, where the high energy
impacts can potentially create additional species with higher
mass, while conventional MS theory assumes that only fragments
with lower mass are produced in the ionizer.
This effect, which generates H2
(mass 2) from water in the analyte of mass spectrometers,
explains the H2 levels in residual gas instead of
presuming it to be unexplained "background" and also explains
the many incorrect H2 analyses of fluid inclusion
volatiles.
It is unsafe to trust H2 analyses of fluid
inclusion volatiles performed by mass spectrometry as H2
is a spurious by-product of the
electron impact ionizer when operating under
"wet" conditions.
 Applied Mineral Exploration
Applied Mineral Exploration Discussion and research relevant to mineral
exploration.
Discussion and research relevant to mineral
exploration. 